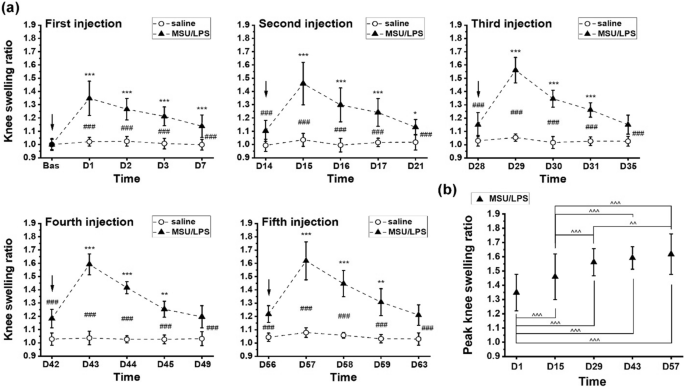
Degenerative joint disease induced by repeated intra-articular injections of monosodium urate crystals in rats as investigated by translational imaging The objective of this work was to assess the consequences of repeated intra-articular injection of monosodium urate (MSU) crystals with inflammasome priming by lipopolysaccharide (LPS) in order to simulate recurrent bouts of gout in rats. Translational imaging was applied to simultaneously detect and quantify injury in different areas of the knee joint. MSU/LPS induced joint swelling, synovial membrane thickening, fibrosis of the infrapatellar fat pad, tidemark breaching, and cartilage invasion by inflammatory cells. A higher sensitivity to mechanical stimulus was detected in paws of limbs receiving MSU/LPS compared to saline-injected limbs. In MSU/LPS-challenged joints, magnetic resonance imaging (MRI) revealed increased synovial fluid volume in the posterior region of the joint, alterations in the infrapatellar fat pad reflecting a progressive decrease of fat volume and fibrosis formation, and a significant increase in the relaxation time T2 in femoral cartilage, consistent with a reduction of proteoglycan content. MRI also showed cyst formation in the tibia, femur remodeling, and T2 reductions in extensor muscles consistent with fibrosis development. Repeated intra-articular MSU/LPS injections in the rat knee joint induced pathology in multiple tissues and may be a useful means to investigate the relationship between urate crystal deposition and the development of degenerative joint disease. Gout represents a highly prevalent form of arthritis, characterized by recurrent episodes of painful acute inflammatory flares in response to monosodium urate (MSU) crystals that deposit predominantly in peripheral joints and surrounding tissues1. Long-term deposition of MSU crystals can result in joint damage. Moreover, gout and osteoarthritis (OA) often occur concomitantly, and a positive correlation between synovial fluid uric acid and OA has been established2. Nevertheless, it is presently not known whether and how these conditions are pathologically linked3,4,5.Recent advances in the understanding of crystal-induced inflammation provided further support for a shared inflammatory pathway between gout and OA. MSU crystals activate the macrophage innate immune response via the Nacht Domain, leucine-rich repeat, and pyrin domain-containing protein 3 (NALP3) inflammasome, which is required for caspase-1 activation and subsequent interleukin (IL)-1β and IL-18 release6. The implication of IL-1β in the pathogenesis of gout has been confirmed by the successful treatment of patients with severe gout using IL-1β inhibition7. In in vitro studies, either the Toll-like receptor 4 (TLR-4) agonist lipopolysaccharide (LPS) or phorbol myristate acetate had to be used to prime cells for effective MSU crystal-induced IL-1β release6,8. Also in the clinics, TLR signaling and priming of inflammasome have been described as a critical step in the process of gouty flares9,10. IL-1β is considered to play a central role in OA as well11,12, and synovial fluid levels of both IL-1β and IL-18 are strongly associated with OA severity2.Preclinical models play an important role in facilitating research in this area. Previous studies demonstrated that a single intra-articular injection of MSU crystals resulted in gouty-arthritis in mice and rats13,14. However, these studies were limited to the acute phase of the response, with an observation time of up to 2 days only, following crystal administration.In this work, we investigated the long-term effect of repeated injections of MSU crystals in combination with lipopolysaccharide (LPS) into the knee joints of rats every two weeks for a maximum of five administrations, with the aim to stimulate NLRP3 inflammasome activation as previously described6,15. We assessed the swelling response after each injection, paw withdrawal sensitivity to mechanical stimuli using electric von Frey, as well as hard and soft tissue joint changes by magnetic resonance imaging (MRI) and micro-computed tomography (micro-CT). Histology was performed at selected time points for the characterization of pathological features at the cellular level. The objective was to assess the consequences of repeated intra-articular crystal/LPS administration with inflammasome priming in order to simulate recurrent gout bouts in rats, using translational imaging to simultaneously detect and quantify injury in different areas of the knee joint.In vivo experimental procedures followed the Swiss animal welfare regulations. The experimental protocols were approved by the Cantonal Veterinary Office of the City of Basel, Switzerland. The study was performed under the license number BS-1438, approved by the Cantonal Veterinary Office of the City of Basel. Authors complied with the ARRIVE guidelines for animal experimentation.AnimalsFemale Lewis/OrlRj rats (n = 25) from Janvier Laboratories (Le Genest-Saint-Isle, France), 150–180 g or eight weeks of age at the beginning of the study, were used. Rats were housed under standard conditions (12-h light/dark cycle), with standard chow and water provided ad libitum. Upon arrival, rats were allowed two weeks of acclimatization before beginning the experiments.Monosodium urate (MSU) crystalsMSU crystals were prepared according to the method reported by Reber et al.16. Initially, 1.0 g of uric acid was added to 6.0 mL of 1.0 N NaOH and 194 mL of double distilled water and then, the mixture was heated. The pH of this mixture was adjusted to 7.2 with 1.0 N HCl. The mixture was allowed to cool slowly at room temperature and then stored at 4 °C for 24 h. Finally, the monourate crystals were dried after washing and then, the crystals were suspended in sterile saline (100 mg/ml).Induction of gouty arthritisTwenty five rats were used. On day 0, the right knee received 50 µl of a mixture containing 40 mg/ml MSU crystals and 0.1 mg/ml LPS from E. coli (0111:B4, Sigma L2630) in saline, while the left knee of the same animal received 50 µl of saline. The intra-articular injections of MSU/LPS into the right and saline into the left knee were then repeated every two weeks (namely on days 0, 14, 28, 42 and 56). Saline was administered into the left knee to verify whether repeated intra-articular injection of fluid might elicit an inflammatory response in the knee joint. Rats were anesthetized with 3.5% isoflurane (Abbott, Cham, Switzerland)/air for each intra-articular injection. At each of the time points days 14, 28, 42, 56 and 70 five animals were culled for post-mortem analyses. The sacrificed rats had received the last MSU/LPS dose 14 days before being euthanized.A preliminary dose–response was performed to select the MSU and LPS dose which led to a robust swelling and cytokine detection in the synovial fluid 24 h after intra-articular dosing (supplementary Fig. 1). The choices for the doses of MSU and LPS for the dose–response were based on published literature (Marcotti et al13 for MSU, De-Melo et al17 and Ahmad et al18 for LPS).Exclusion criterionA weight loss of more than 20% would lead to an exclusion and early euthanasia of an animal. However, no rat needed to be excluded from the study.Knee swellingKnee diameters were measured using calipers immediately before and again on days 1, 2, 3 and 7 after each intra-articular injection. Right and left knee diameters were determined in the medial–lateral direction, with the caliper positioned perpendicularly to the leg axis. Knee swelling was defined as the ratio between the knee diameter at a given time point and the mean knee diameter at baseline, before any injection.Nociceptive testHind-paw sensitivity was evaluated by measuring the mechanical withdrawal threshold using a handheld electronic von Frey unit (Cat # 38450, Ugo Basile, Gemonio, Italy). An animal was placed in a clear box on an elevated mesh screen (models BIO-STD EVF and BIO-PVF, Bioseb, Vitrolles, France), and allowed to habituate for 15 min before testing. A filament was applied to the plantar surface of each hind paw. The force was increased by increments of 0.1 g force units from zero until paw withdrawal. A transducer comprising a digital timer automatically recorded the force eliciting paw withdrawal and the corresponding response latency to the nearest 0.1 s. The filament was applied five times per paw, separated by a 5-min interval to prevent sensitization, and the threshold was defined as an average of the five withdrawals observed within the trials.ImagingDuring acquisitions animals were anesthetized with isoflurane 1.5–2% in air, administered via a nose cone.MRIPerformed with a Pharmascan 7 Tesla scanner (Bruker, Ettlingen, Germany). A T2-weighted spin-echo sequence with the following parameters was applied: 16 echoes spaced by 11 ms, echo time (TE) from 11 to 176 ms, repetition time 2022 ms, pixel size 0.078 × 0.078 mm, slice thickness 0.48 mm, 8 slices, without and with fat suppression. A volume resonator (Model 1P-T11070V3, Bruker) with 72 mm inner diameter was used for transmission. A two-channel phased array receive-only mouse head surface coil (Model 1P-T11204V3, Bruker) was used for signal reception. Joints receiving MSU/LPS were imaged at all the time points specified. In contrast, joints receiving saline were measured solely at baseline and at day 69.Relaxation time T2 for cartilage, infrapatellar pad and muscle was determined by fitting with GraphPad Prism (version 8.1.2, GraphPad Software, San Diego, CA) the corresponding signals from regions-of-interest (ROIs) placed in these anatomical areas as function of TE. Volumes of effusion were determined by segmenting the corresponding signals by their intensity using a region grower algorithm available at the scanner software.Micro-CTMeasurements were performed using a vivaCT-40 micro-CT system (Scanco Medical, Brüttisellen, Switzerland). The scan parameters were: voxel size 17.5 × 17.5×17.5 μm, 426 slices, integration time 130 ms, high resolution, 55 E(kVp), 145 μA, 8 W mode, cone beam continuous rotation.Ex vivo analysesTwo weeks after one or more MSU/LPS injections, rats were euthanized, and synovial fluid collected from the right and left knees. To recover synovial fluid from the joints, the skin was cut vertically to expose the knee joint. The tissue over the knee was carefully opened using scissors and forceps, and the surrounding tissue and patella tendon over the knee was held to form a pocket over the center of the knee. Saline (20 µl) was injected into the knee joint, followed by massaging of the joint to help distribute the saline and free-up synovial fluid from knee compartments. The capsule was then opened, and lavaged a further two times with saline (20 µl). The fluid within the joint was then extracted using a needle and syringe, centrifuged at 2000×g for 10 min at 4 °C, and the supernatant stored at − 80 °C until further analysis. The skin was removed and knees were excised for histology.Multiplex ELISASynovial fluid samples were analyzed using a multiplex enzyme-linked immunosorbent assay (ELISA) with rat specific reagents, following the manufacturer’s protocols (Bio-Rad Laboratories, Hercules, CA). Cytokines tested were interleukin-1α (IL-1α), IL-1β, keratinocyte chemoattractant/growth-regulated oncogene (KC/GRO), macrophage inflammatory protein-1α (MIP-1α), monocyte chemoattractant protein-1 (MCP-1), and vascular endothelial growth factor (VEGF). The dedicated Bio-Plex Manager™ software running on a Bioplex 200 System array reader (Bio-Rad) was used to determine individual concentrations. Because of the small amount of synovial fluid drawn from each animal, data from different time points during the course of the study were pooled to allow statistical comparisons between saline- and MSU/LPS-injected joints.Histological preparationKnee joints were fixed in 10% neutral buffered formalin for three days and then placed in a decalcification solution (ImmunoCal Cat # 1440, Decal Chemical Corp, Suffern, NY) for 5 days. On the fourth day, knees were trimmed along the sagittal axis approximately at the mid-trochlear level to separate medial and lateral condyles of the joint, and decalcification was pursued until completion. After sample dehydration and paraffin embedding, 5-µm-thick sections were cut on the lateral condyle and stained: hematoxylin and eosin (H&E) for the characterization of synovium and proteoglycan-containing cartilage identified by Safranin O/Fast green using a procedure adapted from Lillie and Fulmer19 and from Prophet et al20. Macrophages and osteoclasts were detected with an anti-CD68 antibody (MCA341R, Serotec, Puchheim, Germany) applied on paraffin sections as described by Damoiseaux et al21.Analyses of histological changesHistological assessment was performed on the patello-femoral cartilage (PFC), the anterior femoral cartilage (AFC) and the anterior tibial cartilage (ATC). Joint pathology was scored according to the Mankin system22, considering cartilage surface integrity (0–6), proteoglycan loss (0–4), chondrocyte morphology (0–3), fibrovascular replacement of subchondral marrow fat spaces (0–1), synovitis (0–3) and tidemark breaching (0–1). This methodology was adapted from Takahashi et al23,24. All histological scores were determined by one blinded and trained observer (N.A.).StatisticsMultiplex assay data from synovial fluid samples were analyzed using Student’s t-tests (Origin 2021, OriginLab Corporation, Northampton, MA, USA). Paw withdrawal threshold and MRI data were analyzed using ANOVA with random effects (Systat version 13; Systat Software Inc., San Jose, California, USA) to take into account the longitudinal structure of the data. Tukey post-hoc tests were perform
https://www.nature.com/articles/s41598-021-04125-7
Degenerative joint disease induced by repeated intra-articular injections of monosodium urate crystals in rats as investigated by translational imaging